Certara, a global provider of model-informed drug development and regulatory science, announced the launch of Phoenix 8.0.Phoenix is the most advanced, intuitive, and widely-used software for pharmacokinetic (PK), pharmacodynamic (PD) and toxicokinetic (TK) modeling and simulation. Thank You for Your Interest in Phoenix 8.0 To download Phoenix 8.0 please fill out the form to the right. Once you click submit, you must click the download.
Certara University is dedicated to shaping the future of healthcare innovation through modeling and simulation education. Certara University provides modeling and simulation education, skills, and expertise in the global healthcare industry. We recognize that continuing education in both the theory and practice of modeling and simulation in drug development is pivotal to achieving the many benefits of modeling and simulation.
PK/PD Education:
The use of modeling and simulation in drug development has evolved from being a research nicety to a regulatory necessity. Today, modeling and simulation is leveraged across most development programs to understand and optimize key decisions related to safety, efficacy, dosing, special populations, and other labeling requirements. As a pioneer in this field, Certara has provided in-person classroom and customized training on modeling and simulation. Our programs are designed specifically for both the novice and advanced user of our industry premier software Phoenix workbench for managing, analyzing and reporting pharmacokinetic (PK), pharmacodynamics (PD) and toxicokinetic (TK) data.
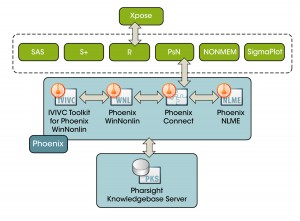
PBPK Education:
Certara Simcyp also provides workshops that focus on the optimal use of compound-specific in vitro and in vivo data, together with system-specific information, to simulate and understand drug behavior in various target populations. This integrated approach informs decisions related to Investigational New Drug (IND) and New Drug Applications (NDA) and assists with the conduct and optimal design of clinical studies. The ultimate aim is to improve the quality of submissions for regulatory approval.
